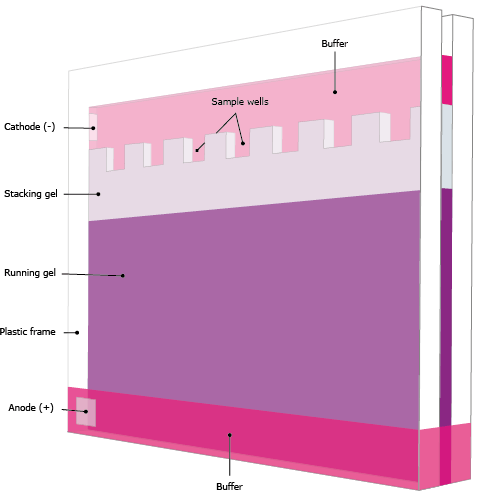
Western blotting is a widely used method for detecting and quantifying specific proteins. However, raw band intensity alone does not always accurately reflect protein levels because variations in sample loading, transfer efficiency, or membrane handling can influence results. Normalization is the process of adjusting the data to account for these variables, ensuring that comparisons between samples are reliable and meaningful.
1. Why Normalization is Important
Without normalization, differences in band intensity could be misleading. For example:
-
Unequal amounts of total protein loaded in each lane
-
Variations in transfer from gel to membrane
-
Differences in antibody binding or exposure times
Normalization corrects for these technical inconsistencies, allowing true biological differences in protein expression to be quantified.
2. Choosing a Normalization Strategy
There are two common strategies for normalization in western blotting:
a. Housekeeping Proteins
Housekeeping proteins are abundantly expressed and generally stable across conditions. Common examples include:
-
β-actin
-
GAPDH
-
Tubulin
Procedure:
-
Detect the target protein and the housekeeping protein on the same blot.
-
Measure the intensity of each band using densitometry software.
-
Divide the target protein’s intensity by the housekeeping protein’s intensity in the same lane.
This ratio provides a normalized expression value, correcting for loading differences.
Tips:
-
Verify that the housekeeping protein expression is stable under experimental conditions; some treatments may alter housekeeping protein levels.
-
Ideally, use multiple housekeeping how western blot works for more robust normalization.
b. Total Protein Normalization
Instead of using a single housekeeping protein, total protein normalization uses the sum of all proteins in each lane. Stains like:
-
Ponceau S
-
Coomassie Brilliant Blue
-
Sypro Ruby
are used to visualize total protein after transfer.
Procedure:
-
Stain the membrane to measure total protein per lane.
-
Quantify total lane intensity using imaging software.
-
Normalize the target protein band intensity by the total protein intensity.
This method is increasingly recommended because it avoids potential variability of housekeeping proteins.
3. Densitometry Analysis
Normalization relies on accurate quantification of band intensities. Steps include:
-
Capture high-quality images of the blot without overexposure.
-
Use image analysis software (e.g., ImageJ) to measure band intensity.
-
Subtract background signal from each measurement to improve accuracy.
-
Apply the normalization method (housekeeping protein or total protein) to calculate adjusted values.
4. Comparing Samples
After normalization, normalized values can be used to:
-
Compare protein expression between control and treatment groups
-
Assess fold changes in protein levels
-
Perform statistical analyses for experimental validation
Example:
If your target protein intensity is 800 arbitrary units and β-actin intensity is 400 units in the same lane, the normalized expression would be 800 ÷ 400 = 2. This value can then be compared across other samples.
5. Best Practices
-
Always include replicates to ensure reproducibility.
-
Validate housekeeping protein stability under your experimental conditions.
-
Keep exposure times consistent to prevent signal saturation.
-
Use appropriate software for densitometry and apply consistent background subtraction.
-
Document all calculations and normalization methods for transparency and reproducibility.
6. Common Pitfalls
-
Using a housekeeping protein that changes with treatment, leading to inaccurate normalization.
-
Overloading or underloading protein samples, which can distort ratios.
-
Ignoring background signal, which may inflate or reduce apparent intensity.
-
Comparing normalized values across different blots without including a standard or reference sample.
Conclusion
Normalizing western blot data is essential for producing reliable, interpretable results. Whether using housekeeping proteins or total protein staining, normalization corrects for technical variability, allowing true biological differences to be assessed. By combining careful experimental design, accurate densitometry, and proper normalization techniques, researchers can confidently quantify protein expression and draw meaningful conclusions from their western blot experiments.
